- 4212 E Los Angeles Ave, PMB #3644, Simi Valley, CA 93063
A new found path, rough and foggy may be the only hope
Clinical Trials, Research & Innovations



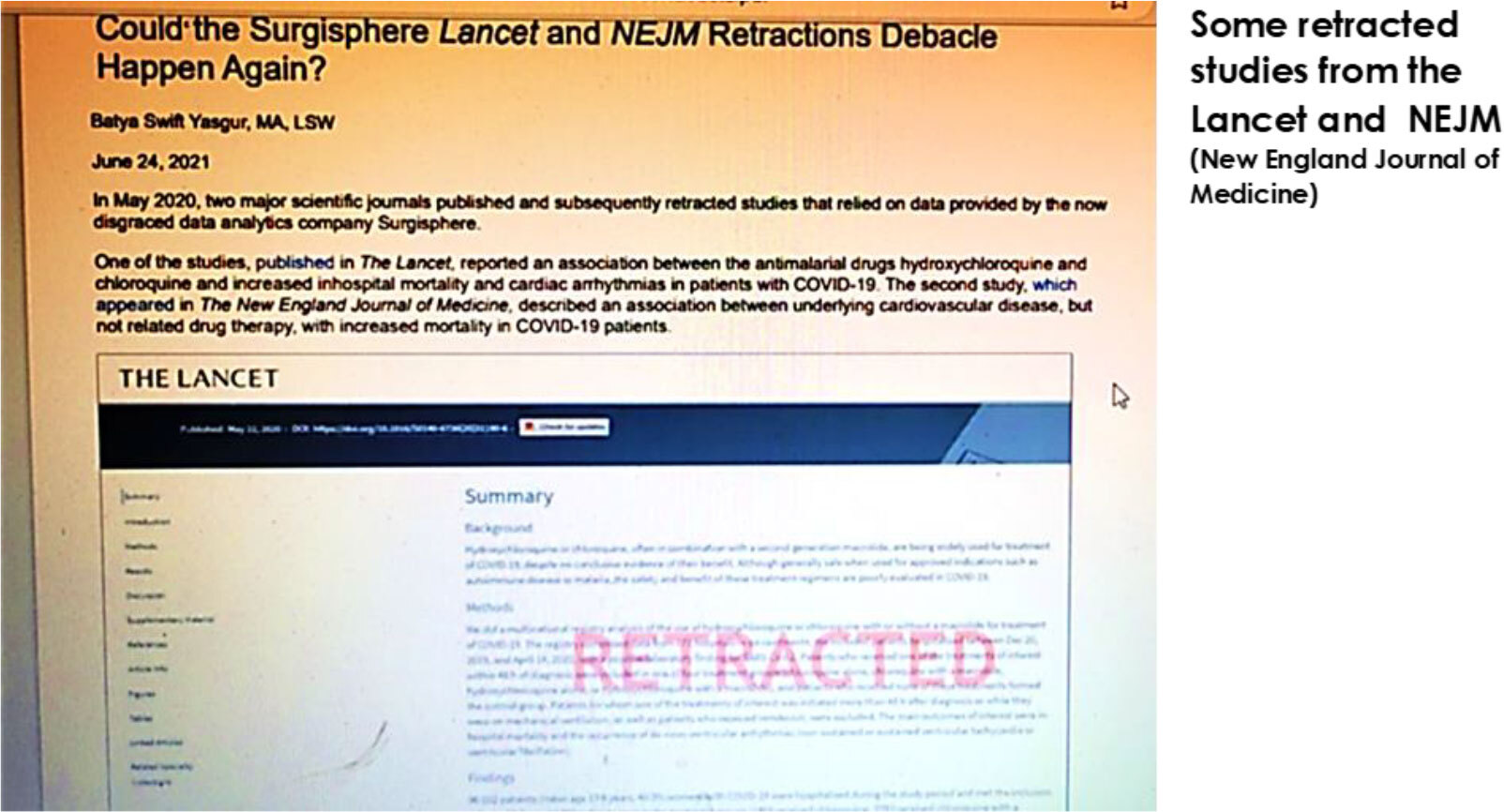
Research studies are the basis for evidence-based medicine, but they are not flawless. While there are inherent problems in conducting research, such as those unavoidable confounding variables, conclusions or interpretations may also disagree, and the researchers working on it are also subject to scrutiny. Study results can be manipulated for various conflicts of interests like when a new drug or product’s FDA approval is at stake, or results can be skewed to gain further grants and continue influx of fundings. Luckily when there is bad work, there is always a countering good work by authorities who are constantly monitoring and eventually retracting flawed studies.
Having been diagnosed with rare or incurable disease, browsing the web can help find many institutions that have ongoing studies on new drugs or treatment approaches on various diseases. One easy way to go is through the ‘Catalogue’ of research online at clinicaltrials.gov. Other authorities and resources are public websites like cdc.gov, nih.gov and NCI’s cancer.gov. Additionally, huge educational healthcare authorities like UCLA Health, USC Keck Medicine, City of Hope, Mayo Clinic, Cleveland Clinic, Hopkins Medicine, Harvard Health and more, have various clinical trials on their respective websites. Before you enroll, talk to a doctor you trust who’s not involved in the research to get an unbiased opinion on what you are to gain or lose. And if you still need further assistance, you can always contact us on the Contact Page, check on Clinical Trials. and write up your issues and / or queries on the Comments & Messages blank box. We can help find your physician trustee.
Take the first step, register to be part of RLabNet research database. It is easy and click on the link below to learn more. There is no obligation nor cost involved.
– Must have had no major health issues in the past 2 years.
– Must have an ailment that is subject of the study
*Research studies usually have few months to two years for recruitment periods and the time frames are posted online during the process. RLabNet Bulletin will have postings of ongoing studies (in-house or outside affiliate) on various subjects of interest.
Research studies are also the roots for innovation. Creating new solutions,new drugs or devices which are fueled by data from clinical research trials and /or experimentations. At times, one does not have to be a rocket scientist or medical expert to create, invent. Enormous information from self- study can also fuel ideas to brilliant innovations!

We love Airborne supplement formulation because it was our same kind of idea-formulation that we contemplated to launch in the early 1990s. We observed remarkable improvement of symptoms on our friends, clients and patients from time to time, using our Airborne-alike supplements in two different forms – as add-up to soups or drinks. Symptoms of colds like headaches, sore throats and runny nose eased up, and improved productivity on coworkers without prolonged call-in sick days. Our excitement went off when the colds supplemental idea came up in the market ahead of us. A teacher thought of the same supplemental idea and introduced her product to Trader Joe’s customers. Nevertheless, Airborne became a subject of our continual observation on how the public would accept the formulation, and Airborne sales went off exponentially. The test of time determines the supplemental formulation’s worth in the market. Though Airborne creator was fined for false-advertising controversy in 2008, Airborne are still sitting on every shelf of pharmaceuticals – big or small. Giant pharmaceuticals came up with their own brands of Airborne-copycats. People buy when they are helped repeatedly.

One out of every four patents is born in California. More than 200,000 patent applications were filed across the 50 states in 2022. California, home to Silicon Valley ‘s tech industry, is the #1 state for patents filed in 2022. It’s the home base for massive companies such as Apple, Google, and Facebook, where multibillion-dollar research budgets buoy innovation. Many states leading the race to patent inventions are also technology hubs. – US Patent and Trademark Office & Census Bureau population estimates
‘New inventions are plenty, but few live up to its novel use ‘
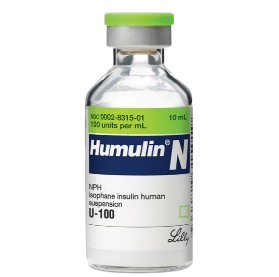
Human Insulin ( Humulin ) is one of the greatest medical, biotech invention of significant novel use. Gone are the days of adverse or allergic reactions from use of insulin derived from pancreases of pigs and cows . Diabetics are in a better place after Humulin, a bioengineered human insulin was created by City Of Hope researchers -inventors : Keiichi Itakura, Ph.D. and Arthur Riggs, Ph.D., in 1979.
People used to connote bacteria in the past as negatives or harmful but the fact is we also have ‘friendly bacteria’ not only around us, but also those that co-exist in our bodily system, like the probiotics guys. Itakura and Riggs made use of friendly bacteria like yeast as host to produce the human- alike insulin, by inserting genes code of human insulin to these hosts and ‘kicking’ them to reproduce human insulin. The harvested human insulin ( Humulin-N) is formulated to have a slower onset of action than regular insulin and a longer duration of activity (slightly less than 24 hours). Genentech (the first American biotechnology company) developed this recombinant DNA technology for mass production and Humulin-N was licensed to Eli Lilly by FDA in Oct 1982, for mass distribution. -Wikipedia

 Theranos founder Elizabeth Holmes
Theranos founder Elizabeth Holmes
Theranos’ 4.5 billion worth innovation hype reduced to zero.
Groundbreaking discoveries are exciting, however there are hyped inventions as well. We had been skeptical about Theranos’ hyped mini blood test technology over a decade ago. While it constantly plugged their blood test analyzer could conduct comprehensive blood tests from finger drops of blood, our affiliate scientists and associates just didn’t buy the idea at the time. Majority’s opinion was, it would take another ten years or so, to get something working with precision, using tiny drops of blood. Additionally, Theranos’ patent was taking years for over a decade to process their blood tech idea. Skeptics of the alleged technology proved right when all dominoes fell in chain reaction. Stanford professor John Ioannidis reported in the Journal of the American Medical Association (JAMA) that NO peer-reviewed research from Theranos had been published in medical research literature. University of Toronto Professor Eleftherios Diamandis analyzed and concluded that most of the company’s claims were exaggerated. In October same year- John Carreyrou of The Wall Street Journal (WSJ) reported that Theranos was just using traditional blood testing machines instead of the company’s own hyped Edison devices to run its tests. A whistleblower worker eventually reported the company’s irregularities to the New York State Department of Health. Series of inspection, lawsuits and settlements followed after the WSJ story. Founder Elizabeth Holmes and software engineer Ramesh Balwani were eventually indicted on multiple counts of wire fraud and conspiracy to commit wire fraud. Investors, doctors and patients were defrauded. Holmes was found guilty and was sentenced 11 years and 3 months in prison. On May 30, 2023, she began serving her sentence at Federal Prison Camp, Bryan in Bryan, Texas. -Wikipedia.
Some popular ideas are not patentable for being obvious.

The amazing and daring Betty Hartman in flight with the zipline during her 92nd birthday!
The idea of Zipline is around thousands of years ago as a mode of transporting goods or mode of transportation. Despite the centuries-long history of ziplines with various uses, modern zipline recreational courses can trace their origins back to 1995, when Canadian entrepreneur Darren Hreniuk built the world’s first recreational zipline course in Monteverde, Costa Rica. Although he tried to patent the concept and aggressively block rival course from opening, the courts ultimately ruled that zipline recreational technology are not patentable and belong in the public domain.
Today, 72 countries boast recreational zipline courses. – wikipedia







celebrity cruises.com / courtesy
RLabNet fellow, a previous REACT operative ziplining for fun and thrill during the REACT Sport Fest.
RLabNet currently has more than a dozen of old and new ideas at work especially on healthcare systems. Ideas are conceived from observational studies and continuing medical education. While some devices or algorithms at work will help avoid medical errors, misdiagnosis, or improve lab techniques and treatment approaches; some wares and devices may unfortunately render some current systems or jobs obsolete.